Solar Energy Efficiency: Beyond the Shockley Queisser Limit
The Shockley Queisser Efficiency Limit
 The concept of the Shockley Queisser
Efficiency Limit was first formulated by William Shockley and Hans Queisser in
1961. It represents the maximum efficiency that any single-junction solar cell
can achieve, typically around 33% under standard test conditions (STC). These
conditions simulate solar noon during the spring and autumn equinoxes in the
continental United States, with the solar cell directly facing the sun.
The concept of the Shockley Queisser
Efficiency Limit was first formulated by William Shockley and Hans Queisser in
1961. It represents the maximum efficiency that any single-junction solar cell
can achieve, typically around 33% under standard test conditions (STC). These
conditions simulate solar noon during the spring and autumn equinoxes in the
continental United States, with the solar cell directly facing the sun.
Initially, Shockley and Queisser calculated a limit of 30% for silicon solar cells. However, modern calculations have refined this to 33% for any single-junction solar cell. Despite advancements, current solar cell production efficiencies vary based on the semiconductor material's band gap.
Sun power reported the highest efficiency for silicon cells in 2012, reaching 24% at the cell level and 20% at the module level. Meanwhile, the University of New South Wales in Sydney, Australia, achieved a record efficiency of 25% in laboratory settings. Despite ongoing efforts to surpass the SQ Limit, it remains a significant benchmark, applicable to nearly all solar cells on the market, with only a fraction of exceptions.
The Critical SQ Limit Assumptions:
- One semiconductor material (excluding doping materials) per solar cell.
-
One p/n junction per solar cell.
-
The sunlight is not concentrated-a "one sun" source.
- All energy is converted to heat from photons greater than the band gap.
Where Does The 67% Of Energy Loss Go?
- 47% of the solar energy gets converted to heat.
-
18% of the photons pass through the solar cell.
-
02% of energy is lost from local recombination of newly created
holes and electrons.
-
33% of the sun's energy is theoretically converted to electricity.
- 100% total sun's energy.
If the theoretical limit for silicon cells is about 30%, what happens to the other 6% that is lost from the best production cell efficiency of 24%? Some sunlight is always reflected off the surface of the cell even though the surface is usually texturized and coated with an anti-reflective coating. In addition, there are some losses at the junction of the silicon cell with the electrical contacts that carry the current to the load. Finally, there are some losses due to manufacturing impurities in the silicon.
What Electro-Magnetic Waves Are Absorbed by a Solar Cell?
A solar cell absorbs electromagnetic waves selectively, primarily those within the visible spectrum and a portion of the near-infrared spectrum. The energy carried by these photons must meet or exceed the band gap energy of the semiconductor material within the solar cell for electron excitation to occur. Photon absorption occurs when the energy of a photon matches or exceeds the band gap energy, allowing electrons to transition from the valence band to the conduction band, creating an electric current. However, photons with energy below the band gap simply pass through the solar cell without inducing electron excitation.
In practical terms, photons within the visible spectrum are generally absorbed by solar cells, as they possess sufficient energy to promote electron movement across the band gap. Conversely, infrared, microwave, and radio waves lack the necessary energy and pass through the solar cell without contributing to electricity generation.
On the other end of the spectrum, X-rays and gamma rays have energies far exceeding the band gap energy and are not absorbed by the solar cell at all. While photons with excess energy beyond the band gap can still create electron-hole pairs, the surplus energy is dissipated as heat rather than contributing to electrical output. This phenomenon illustrates the importance of matching the band gap energy with the energy of incoming photons to optimize solar cell efficiency.
Strategies to Exceed the SQ Limit
Basically, the strategies to obtain better efficiencies than the SQ Limit predicts are to work-around one or more of the critical assumptions listed above (and shown again below).
- One semiconductor material (excluding doping materials) per solar cell. Use more than one semiconductor material in a cell.
- One P/N junction per solar cell. Use more than one junction in a cell - "tandem cells".
- The sunlight is not concentrated - a "one sun" source. Sunlight can be concentrated about 500 times using inexpensive lenses.
- All energy is converted to heat from photons greater than the band gap. Combine a PV semiconductor with a heat-based technology to harvest both forms of energy and/or use "quantum dots" to harvest some of the excess photon energy for electricity.
Strategies 1) and 2) Multi-junction Solar Cells - "Tandem Cells"
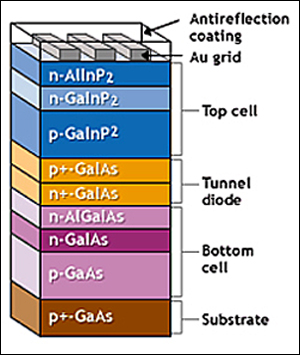 The earliest and most frequent work around
to the SQ Limit has been the use of multiple p/n junctions, each one tuned to a
different frequency of the solar spectrum. Since sunlight will only react
strongly with band gaps roughly the same width as their wavelength, the top
layers are made very thin so they are almost transparent to longer wavelengths.
This allows the junctions to be stacked, with the layers capturing the shortest
wavelengths on top, and the longer wavelength photons passing through them to
the lower layers.
The earliest and most frequent work around
to the SQ Limit has been the use of multiple p/n junctions, each one tuned to a
different frequency of the solar spectrum. Since sunlight will only react
strongly with band gaps roughly the same width as their wavelength, the top
layers are made very thin so they are almost transparent to longer wavelengths.
This allows the junctions to be stacked, with the layers capturing the shortest
wavelengths on top, and the longer wavelength photons passing through them to
the lower layers.
The example of a multi-junction cell on the right has a top cell of gallium indium phosphide, then a "tunnel diode junction", and a bottom cell of gallium arsenide. The tunnel junction allows the electrons to flow between the cells and keeps the electric fields of the two cells separate. Most of today's research in multi-junction cells focuses on gallium arsenide as one of the component cells as it has a very desirable band gap. Performing a calculation using the SQ methodology; a two-layer cell can reach a maximum theoretical efficiency of 42% and three-layer cells 49%.
The record for a multi-junction cell is held by the University of New South Wales (UNSW) in Sydney, Australia at 43% using a five-cell tandem approach. However, the UNSW tandem cell is very expensive. In addition to the cost issue, there are other constraints that make the tandem cells complex. For example, all the layers must be lattice compatible with one another in their crystalline structure and the currents from each individual cell must match the other cells. Multi-junction cells are commercially used in only special applications because their expense currently outweighs any efficiency improvement. At the moment they are used in space where weight is most important and in concentrated PV systems where the sunlight is focused on a very small cell area requiring only small amounts of semiconductors per cell.
Strategy 3) Concentrate the Sunlight
Concentrated Photovoltaics (CPV), in which sunlight is focused onto a small solar cell by lenses to generate more power per unit of surface area, was an early favorite to increase solar efficiency. CPV’s main attraction is that it can leverage modest "one sun" cell electricity production to a much larger scale production using relatively simple and inexpensive optical concentration.
 Instead of a typical 6 inch by 6-inch solar
cell, a 7 inch by 7-inch square plastic Fresnel (pronounced Fray-NELL) lens incorporating
circular facets, is used to focus the sunlight as shown on the right. A tiny,
39% efficient multi-junction solar cell is mounted at the focal point which
converts the sun's energy into electricity. Future cell efficiencies are
expected to approach 50%. The Fresnel lens concentrates the sun's energy about
500 times its normal intensity. A number of Fresnel lenses are manufactured as
a single plastic piece. The tiny solar cells are mounted on a supporting plate
at locations corresponding to the focus point of each Fresnel lens. Hundreds of
lenses make up a solar array mounted on a sun tracking heliostat. With a high
"module efficiency" of 31%, CPV systems take up less land than
traditional PV systems, use no water, and are ideal for desert type areas.
Instead of a typical 6 inch by 6-inch solar
cell, a 7 inch by 7-inch square plastic Fresnel (pronounced Fray-NELL) lens incorporating
circular facets, is used to focus the sunlight as shown on the right. A tiny,
39% efficient multi-junction solar cell is mounted at the focal point which
converts the sun's energy into electricity. Future cell efficiencies are
expected to approach 50%. The Fresnel lens concentrates the sun's energy about
500 times its normal intensity. A number of Fresnel lenses are manufactured as
a single plastic piece. The tiny solar cells are mounted on a supporting plate
at locations corresponding to the focus point of each Fresnel lens. Hundreds of
lenses make up a solar array mounted on a sun tracking heliostat. With a high
"module efficiency" of 31%, CPV systems take up less land than
traditional PV systems, use no water, and are ideal for desert type areas.
Despite the concentration advantages, CPV has been slow to gain market share. While the tiny solar cells use less of the expensive semiconductor materials, cost is a factor as a two-axis sun tracking heliostat is necessary to accurately keep the focus point on the solar cell as the sun travels east to west each day and north and south each season. CPV does not do well in cloudy climates as diffuse sunlight does not concentrate well. In addition, the large heliostats are not well suited for the small installations that have been the mainstream of the recent PV market. Today, CPV costs are very competitive and CPV is benefiting from growing demand for large utility size solar plants, especially in the desert areas of California, Arizona, Spain, and Australia.
Strategy 4a) Combine a PV cell with heat-based technology (PETE)
 Pete Diagram the Stanford University Photon
Enhanced Thermionic Emission (PETE) prototype uses concentrated sunlight as its
source of energy and in a two-step process uses both the sun's photon energy
and its heat. A thermionic converter consists of two electrodes separated by a
vacuum, see the figure to the right. When the cathode is heated to a high
temperature, electrons become excited, jump across the thin vacuum to the
relatively cold anode, and drive a current through an external circuit back to
the cathode. In the Stanford prototype, the cathode emitter is a semiconductor
material rather than a metal electrode. First, the highly concentrated sunlight
photons partially excite the electrons in the cathode semiconductor so that in
step two, the remaining heat energy necessary for emission is lower than that
for a standard thermionic converter. The surface of the cathode on the vacuum
side is texturized to increase emissions. PETE converts about 25% of the
sunlight's energy into electricity at 200°C and higher efficiencies at higher
temperatures, i.e. 45% at 1000°C. Because of the high temperatures this type of
solar system would probably only be used by utilities to generate grid
electricity. A lot of work needs to be done to get from today's laboratory set
up to a production product in the field. A competitive product is probably 8 to
10 years away.
Pete Diagram the Stanford University Photon
Enhanced Thermionic Emission (PETE) prototype uses concentrated sunlight as its
source of energy and in a two-step process uses both the sun's photon energy
and its heat. A thermionic converter consists of two electrodes separated by a
vacuum, see the figure to the right. When the cathode is heated to a high
temperature, electrons become excited, jump across the thin vacuum to the
relatively cold anode, and drive a current through an external circuit back to
the cathode. In the Stanford prototype, the cathode emitter is a semiconductor
material rather than a metal electrode. First, the highly concentrated sunlight
photons partially excite the electrons in the cathode semiconductor so that in
step two, the remaining heat energy necessary for emission is lower than that
for a standard thermionic converter. The surface of the cathode on the vacuum
side is texturized to increase emissions. PETE converts about 25% of the
sunlight's energy into electricity at 200°C and higher efficiencies at higher
temperatures, i.e. 45% at 1000°C. Because of the high temperatures this type of
solar system would probably only be used by utilities to generate grid
electricity. A lot of work needs to be done to get from today's laboratory set
up to a production product in the field. A competitive product is probably 8 to
10 years away.
Strategy 4b) Quantum Dots Absorb Excess Photon Energy
In a regular solar cell, each photon collision generates a particle pair consisting of one free hole and one free electron. Quantum Dots are extremely small "nanocrystals" (the names are used somewhat interchangeably) interspersed in a larger semiconducting material. Quantum Dots (QDs) range between 1 and 20 nanometers in size (one nanometer is one billionth of a meter). Semiconductors at this size have different physical properties than their big brothers. When photons with energy greater than the band gap energy collide with a Quantum Dot several "hot" hole/electron pairs can be created as opposed to one pair and heat. Although silicon can be used as a nanocrystal, lead selenide (PbSE) also a semiconductor, is being used more frequently as the material of choice.
Another characteristic of a Quantum Dot is that different sizes capture different wavelengths of light. Small dots capture small wavelengths and larger dots bigger wavelengths. Some researchers have figured out how to stack the dots from small to large to capture more photon energy similar to how tandem cells do. Once a hot electron is created inside a Quantum Dot, it stretches its lifetime as much as a 1000 times before it cools. The electrons like to stay inside the QD. One of the challenges was to figure out how to extract the hot electrons from the QDs. No solar cells produced prior to December, 2011 have quantum efficiencies greater than 100 percent. Quantum efficiency (not to be confused with solar cell efficiency) per the National Renewable Energy Laboratory (NREL) located in Boulder, Colorado, is the “ratio of collected charge carriers (electrons or electron holes) to incident photons”. In layman terms - it’s the ratio of the number of electrons produced in a solar cell to the number of the sun's photons hitting the cell.
Researchers from the NREL have reported quantum efficiencies of 114 percent in solar cells “excited” from photons from the high-energy region of the solar spectrum. That is from the near ultraviolet through the visible light spectrum - 350 to 700 nanometers. Energy is always conserved. The extra electrons come from the extra energy left over after the initial photon-electron collision. Light waves below 700 nanometers do not have enough energy to dislodge more than one electron-hole pair. NREL achieved this result with a layered quantum dot "experimental cell" composed of a surface of anti-reflective glass, a thin layer of semiconductor zinc oxide “textured” at the nano level, a QD layer of lead selenide doped with ethanedithol (a bonding agent) andhydrazine (a deposition stabilizer), and a thin layer of gold for the collector electrode. This process, which creates more than one electron-hole pair from a single photon, is called "multiple exciton generation" (MEG) by NREL.
The practical upper limit for "thin film" solar cells is thought to be about 20%. The upper limit using Quantum Dots is thought to be about 30%. It should be emphasized that the research into Quantum Dots is at a very basic stage of demonstrating scientific principles. No one at this time has actually made a pre-production Quantum Dot solar cell. Production solar cells using Quantum Dots are thought to be about 10 years into the future.Post a Comment:
You may also like:

